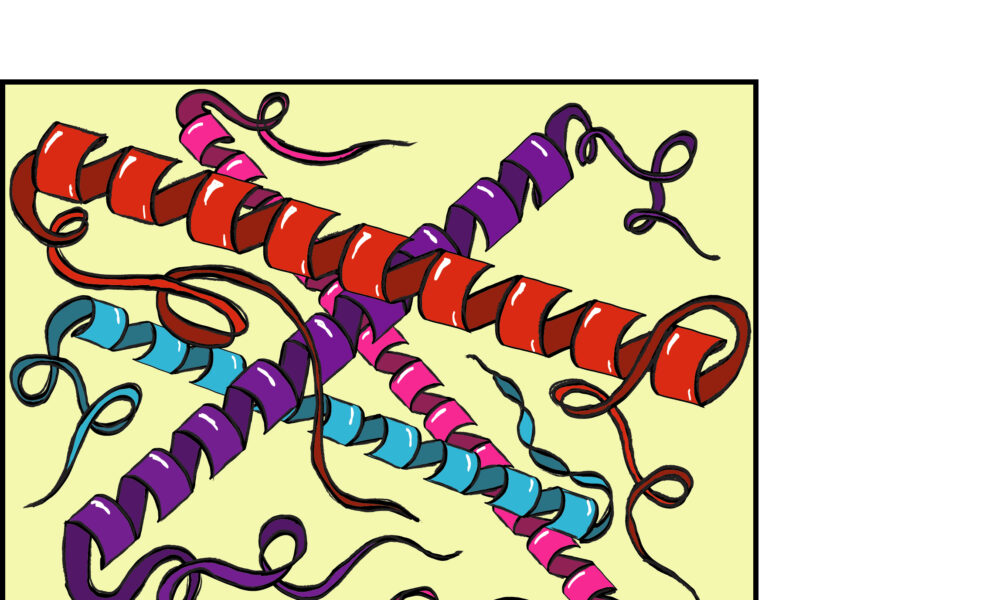
Proteins are the workhorses of our cells, responsible for a vast array of functions that keep us alive and healthy. From building muscle to fighting disease, their intricate shapes determine their specific roles. Although the task has so far eluded scientists, they hope to ultimately incorporate lab-designed proteins into personalized medicine that anyone can take for any condition.
For decades, scientists have been grappling with a fundamental mystery: How to accurately predict a protein’s folding pattern based solely on the string of amino acids that comprise it. This challenge, known as the protein folding problem, has long been a hurdle in developing new drugs and therapies, as experimentally determining shape costs labs considerable time, money, and effort.
However, a revolution in protein-folding prediction is underway, driven by the power of artificial intelligence (AI). Pioneering tools are making significant strides in predicting protein structures with unprecedented accuracy. These tools include AlphaFold, developed by Google DeepMind, and RoseTTAfold, developed by the Institute for Protein Design (IPD) at the University of Washington. They use a combination of deep learning algorithms and advanced statistical methods to analyze vast databases of known protein structures, allowing for the identification of patterns and relationships that can then be applied to predict new proteins. This streamlines the initial process of developing protein-based medicines.
For a more in-depth look at the software, Sebastian Kenny, a postdoc at the IPD, wrote about integrating different predictive software they have developed in an email to The Tribune.
“We start with RFDiffusion generating protein backbones around a hotspot of our choosing,” Kenny wrote. “Following that, ProteinMPNN assigns amino acid sequences to the generated backbone. This sequence is inferred from other structures of proteins that have been studied in the past. Finally, we validate and score our designs using RoseTTAfold or AlphaFold to see the likelihood of our designed protein forming the complex we expect.”
Kenny explained that using this technique dramatically shortens the process of protein design and integration, as it now requires only a few weeks to a month to model and validate.
Paul Harrison, associate professor in the Department of Biology at McGill, highlighted the time-saving potential of programs like AlphaFold by explaining how predictive algorithms can accelerate experimentation.
“Firstly, predictive algorithms can cut costs, and secondly they help in generating experimental hypotheses, for example, structures predicted by the AlphaFold algorithm are often as good as an experimentally determined structure derived from X-ray crystallography,” Harrison wrote in an email to The Tribune.
Lucas Elliot, U1 Science, is excited to see how these techniques could be integrated into the university’s curriculum.
“Computer science is something we’re now learning younger and younger, but it’s not standard yet. But I think it will become standard,” Elliot said in an interview with The Tribune. “That makes the way for understanding AI. And I would love to see work with AI tools in my curriculum, as soon as possible. I do think it’ll be a little slow. Especially with very established universities, everyone loves these new tools, and I’m sure all the professors are using them. But then incorporating them in a teaching class is slower.”
It would be beneficial for McGill to strengthen its teaching by integrating open-source tools like AlphaFold and RoseTTAfold into the curriculum. Learning how to use these innovative techniques could significantly accelerate the development of life-saving medicines as graduates contribute to the world as new scientists and engineers.
Jerome Waldispuhl, associate professor in McGill’s School of Computer Science, acknowledged the usefulness of tools like AlphaFold but highlighted the need for analytical thinking.
“It’s a catalyzer. It allows you to explore combinations, and [the] mixing of things you already know much faster, based on current knowledge, but it doesn’t replace human supervision,” Waldispuhl said in an interview with The Tribune.
Waldispuhl emphasized that these algorithms need to have their findings validated by scientists. AI in biology could shave off years of work with faster designs for potential medicines, but we need critical minds to make the best use of it.