In 2020, the Jahani-Asl Lab in the Division of Experimental Medicine at McGill discovered that gene therapy techniques could make certain brain tumours more receptive to radiation treatment. Since then, the team has made yet another groundbreaking advancement—identifying a protein pathway that, when suppressed, could lead to reduced tumour growth.
Glioblastoma is the most severe of all brain tumours; the prognosis is often grim, and treatment typically requires surgery, and a combination of chemotherapy and ionizing radiation. Brain tumour stem cells (BTSCs) are notoriously difficult to manage due to their capacity to self-renew and proliferate.
“Despite these efforts, there are […] brain tumour stem cells that resist therapy and stay in a dormant state […] and then after a while, they regrow and they form a tumour,” Ahmed Sharanek, a postdoctoral researcher and co-lead author of the study, said in an interview with The McGill Tribune. “Finding a therapy that specifically targets these brain tumour stem cells will be a great advance in glioblastoma therapy.”
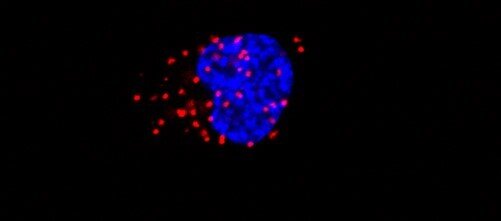
In a recent study published in Cell Reports, the researchers discovered that the production of galectin-1, a carbohydrate-binding protein, is under the control of the EGFR/STAT3 signalling pathway—a pathway known to promote tumourigenesis. When galectin-1 is being produced, it forms a complex with a transcription factor called HOXA5, and together, they send signals to brain tumour stem cells to stimulate growth.
Sharanek explained how the researchers performed next-generation sequencing and biochemical assays to understand the role of galectin-1 in the oncogenic pathway.
“We tried to tackle the glioblastoma tumours that are dependent on the EGFRvIII pathway,” Sharanek said. “We did this because it is a major pathway that occurs in 40 per cent of tumours.”
A majority of glioblastomas express mutations in the EGFR gene that activates STAT3—and though researchers have been trying to suppress this pathway, they have been largely unsuccessful. The next step was to target galectin-1 and to prevent it from binding to HOXA5. Using CRISPR gene editing and pharmacological techniques, the researchers inhibited the expression of this protein in the cells of animal models to observe its effect on tumour growth.
Given their understanding of the pathway’s mechanism, Sharanek and his colleagues expected there to be reduced tumour growth in the experimental mice that received the galectin-1 manipulation. But what happened was even more promising—for several months, the tumours did not grow at all. In fact, the mice who received treatment lived for 12 months, a significant lifespan by laboratory standards.
“[It was] really startling for us,” Sharanek said. “When we deleted this protein in brain tumour cells and injected them in mouse models, we thought that we were going to decrease the growth of tumours to a certain extent, but what we observed […] was that they simply did not [grow].”
The results were encouraging, especially given that much of the lab work was slowed by pandemic restrictions.
The paper released last year detailed how the deletion of a mitochondrial gene called Oncostatin M could make stem cells easier to target with radiation. Since the galectin-1 protein is involved in a separate oncogenic pathway, Sharanek is hopeful that multiple points of attack could improve patients’ responsiveness to radiation therapy.
“In the response of glioblastoma tumours to therapy, these cells are very smart,” Sharanek said. “If you hit one pathway, […] the [tumours] adopt another pathway to rescue themselves. So if you are hitting two major pathways at the same time, your chance [of eradicating] them is much higher.”
With galectin-1 suppression as a new weapon in the arsenal against tumour cells, along with the lab’s previous work on gene therapy, the lab continues to make strides in the world’s understanding of glioblastoma.